 Greater value of bond order for H 2 molecule than H 2 + ion shows that two H 2 molecule is more stable than H 2 +.īond length of H 2 is smaller than that of H 2 + ion.Īs no unpaired electron is present, the H 2 molecule should be diamagnetic. Positive value of bond order indicates that H 2 molecule is stable.īond order value of 1 means that two hydrogen atoms are connected by a single bond. The electronic configuration of H 2 is ( σ(1s) ) 2 The electronic configuration of H 2 + is ( σ(1s) ) 1Ģ) Positive bond order means it is stable.ģ) One unpaired electron is present. Greater the number of unpaired electrons present in the molecular or ion, greater is its paramagnetic nature.Įlectronic configuration of Homonuclear Diatomic Molecules If the molecules has some unpaired electrons ,it is paramagnetic in nature. If all the electrons in the molecule are paired, it is diamagnetic in nature. Greater the bond order, shorter is the bond length.Ħ) Diamagnetic and paramagnetic nature of the molecules the bond order is negative or zero.ģ) Relative stability of molecule in terms of bond orderįor diatomic molecules ,the stability is directly proportional to the bond order.Ī molecule with the bond order of 3 is more stable than a molecule with bond order of 2 and so on.Ĥ) Nature of bond in terms of bond order :īond order 1 ,2 and 3 mean single ,double and triple bond.īond length is found to be inversely proportional to the bond order. The molecule is unstable if N b < Na i.e. Σ(1s) Na ,the molecule is stable because greater number of bonding orbitals are occupied than antibonding orbital, resulting in a net force of attraction.Ģ) If N b Na ie. 
The first ten molecular orbitals may be arranged in order of energy as follow: Combining the in-phase orbitals results in a bonding orbital.Energy level diagram for Molecular orbitals One contains the axis, and one contains the perpendicular. Combining the out-of-phase orbitals results in an antibonding molecular orbital with two nodes. Side-by-side overlap of each two p orbitals results in the formation of two π molecular orbitals. For the out-of-phase combination, there are two nodal planes created, one along the internuclear axis and a perpendicular one between the nuclei.įigure 7.7.6. Electrons in this orbital interact with both nuclei and help hold the two atoms together, making it a bonding orbital. In molecular orbital theory, we describe the \pi orbital by this same shape, and a \pi bond exists when this orbital contains electrons. In valence bond theory, we describe π bonds as containing a nodal plane containing the internuclear axis and perpendicular to the lobes of the p–\pi orbitals, with electron density on either side of the node. 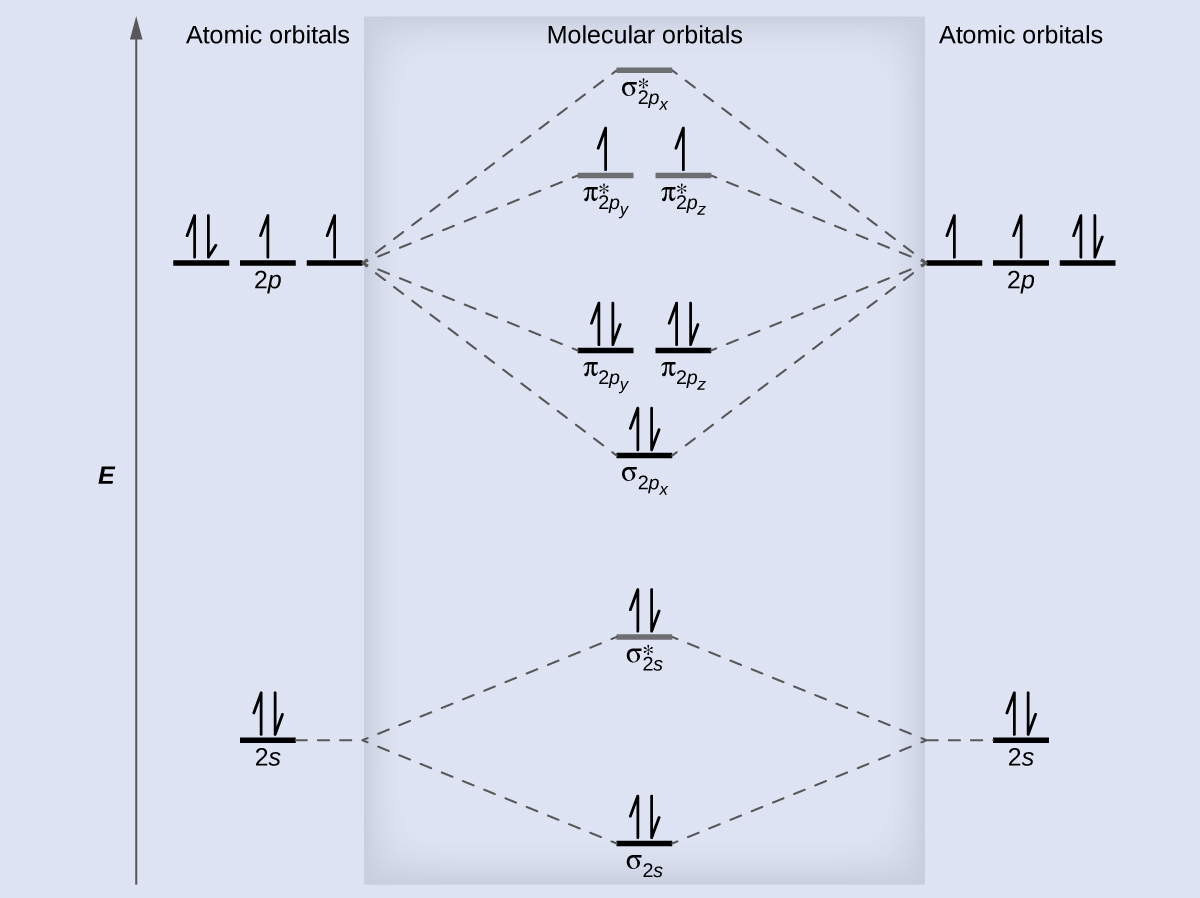
The side-by-side overlap of two p orbitals gives rise to a pi (\pi) bonding molecular orbital and a \pi* antibonding molecular orbital, as shown in Figure 7.7.6. Combining wave functions of two p atomic orbitals along the internuclear axis creates two molecular orbitals, σp and σ∗p. Just as with s-orbital overlap, the asterisk indicates the orbital with a node between the nuclei, which is a higher-energy, antibonding orbital.įigure 7.7.5. There is an \ce^* (antibonding) (read as “sigma-p-x” and “sigma-p-x star,” respectively). This electronic structure adheres to all the rules governing Lewis theory. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
